PharmaMar Has Announced That the Spanish Medicines Agency Has Authorized the APLICOV-PC Clinical Trial With Aplidin® (Plitidepsin) for the Treatment of Patients With COVID-19

The Spanish Medicines Agency authorizes the Spanish Hipra vaccine to go to the last phase of the trial - Spain's News
/cloudfront-eu-central-1.images.arcpublishing.com/prisa/6HWW7462G2CKUA7TELJDT6HZZ4.jpg)
Covid-19 vaccine in Spain: Spain will restart AstraZeneca shots next week after European Medicines Agency finds vaccine 'effective and safe' | Society | EL PAÍS English

Spanish Agency for Medicines and Health Products Expedites Drug National Code Application Process - HealthEconomics.Com
![La Moncloa. 12/01/2021. First doses of Moderna vaccine against COVID-19 arrive in Spain [Government/News] La Moncloa. 12/01/2021. First doses of Moderna vaccine against COVID-19 arrive in Spain [Government/News]](https://www.lamoncloa.gob.es/serviciosdeprensa/notasprensa/sanidad14/PublishingImages/2021/120121-moderna_covid-19.jpg?RenditionID=33)
La Moncloa. 12/01/2021. First doses of Moderna vaccine against COVID-19 arrive in Spain [Government/News]

Evaluation of new medicines in Spain and comparison with other European countries | Gaceta Sanitaria
Instruction document of the Spanish Agency of Medicines and Medical Devices for conducting clinical trials in Spain
DIRECTRICES DE LA AGENCIA ESPAOLA DE MEDICAMENTOS Y PRODUCTOS SANITARIOS SOBRE LA NOMENCLATURA DE LAS SUSTANCIAS ACTIVAS DE LOS

Spanish Medicines Agency issues health alert and withdraws some popular high blood pressure pills | Sur in English
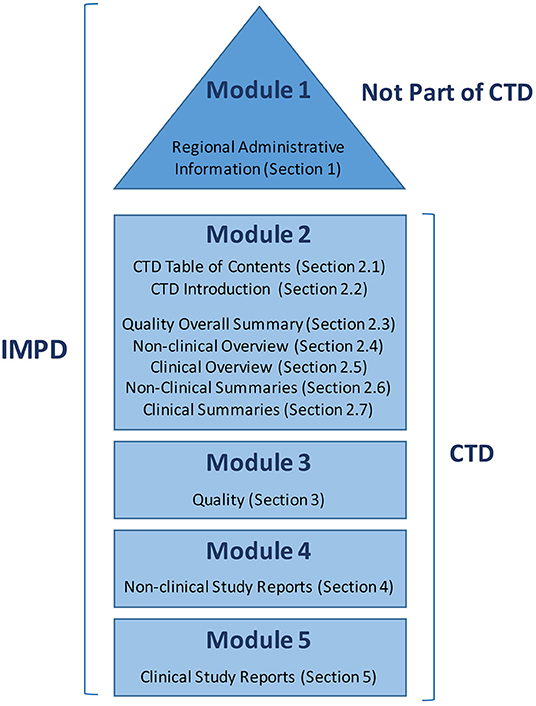
Frontiers | Transitioning From Preclinical Evidence to Advanced Therapy Medicinal Product: A Spanish Experience