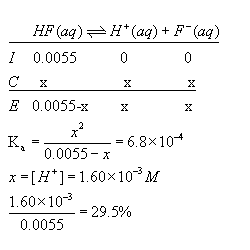![Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download](https://slideplayer.com/4572254/15/images/slide_1.jpg)
Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download

SOLVED: (a) Calculate the percent ionization of a 0.20 M solution of the monoprotic acetylsalicylic acid (aspirin) for which Ka=3.0 × 10^-4. (b) The pH of gastric juice in the stomach of

SOLVED: Calculate the percent ionization of a 0.10 M solution of acetic acid (CH3COOH) with a pH of 2.89.

SOLVED: a What is the percent ionization in 1.1 M NH: Kb = 1.8 x 10 75)2 Percent ionization % b What is the percent ionization in 0.090 M NH: Kb =
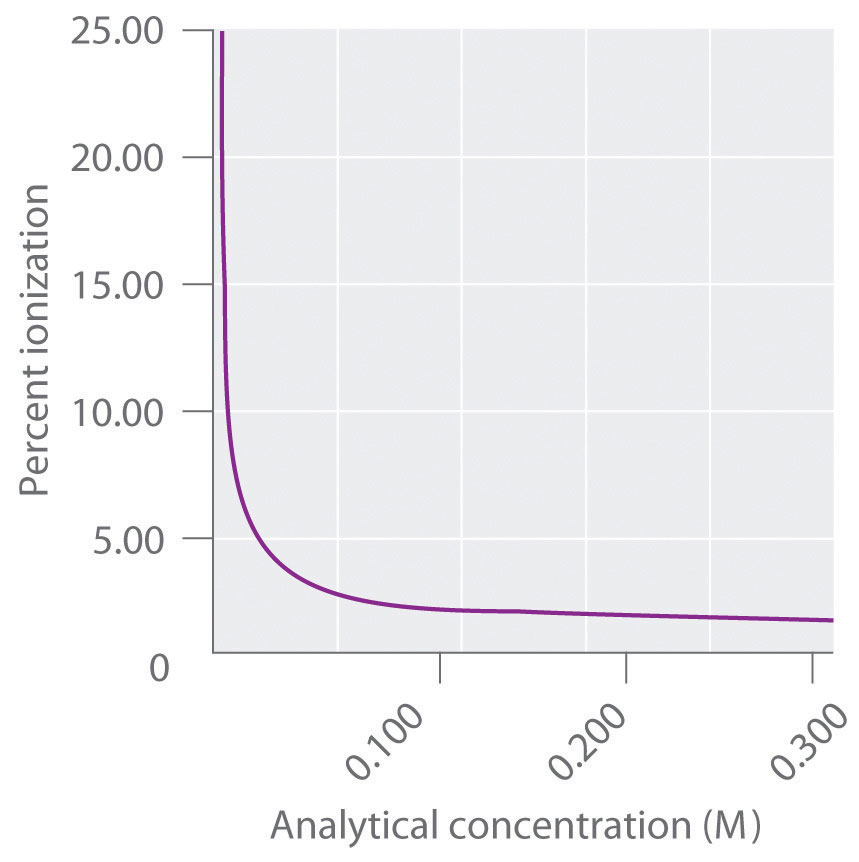
![Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download](https://images.slideplayer.com/15/4572254/slides/slide_3.jpg)
Acids and Bases Calculating Percent Ionization Percent Ionization = 100 In this example [H 3 O + ] eq = 4.2 10 −3 M [HCOOH] initial = 0.10 M [H 3 O. - ppt download


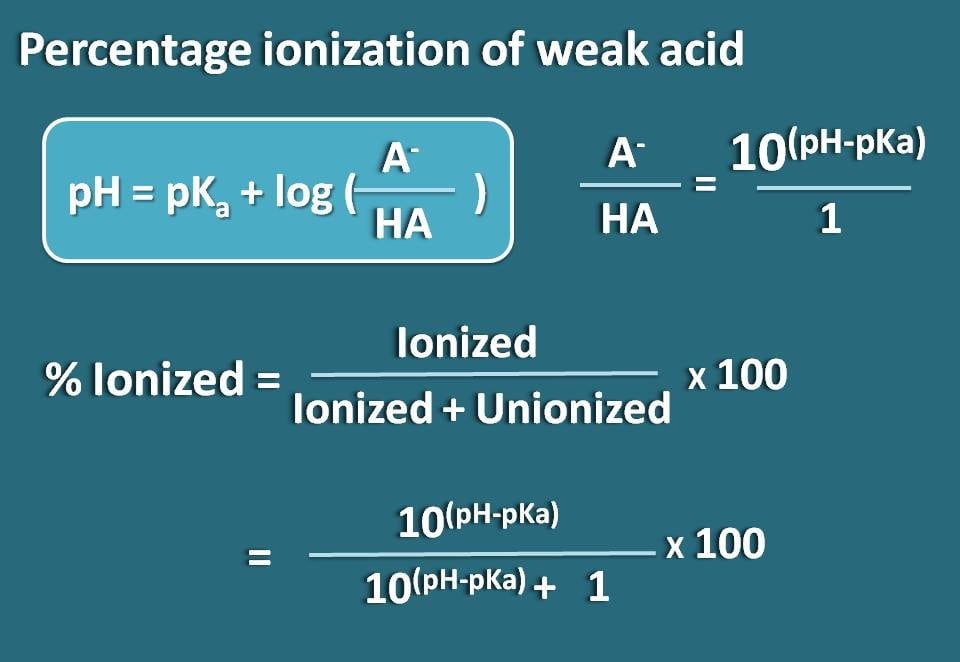



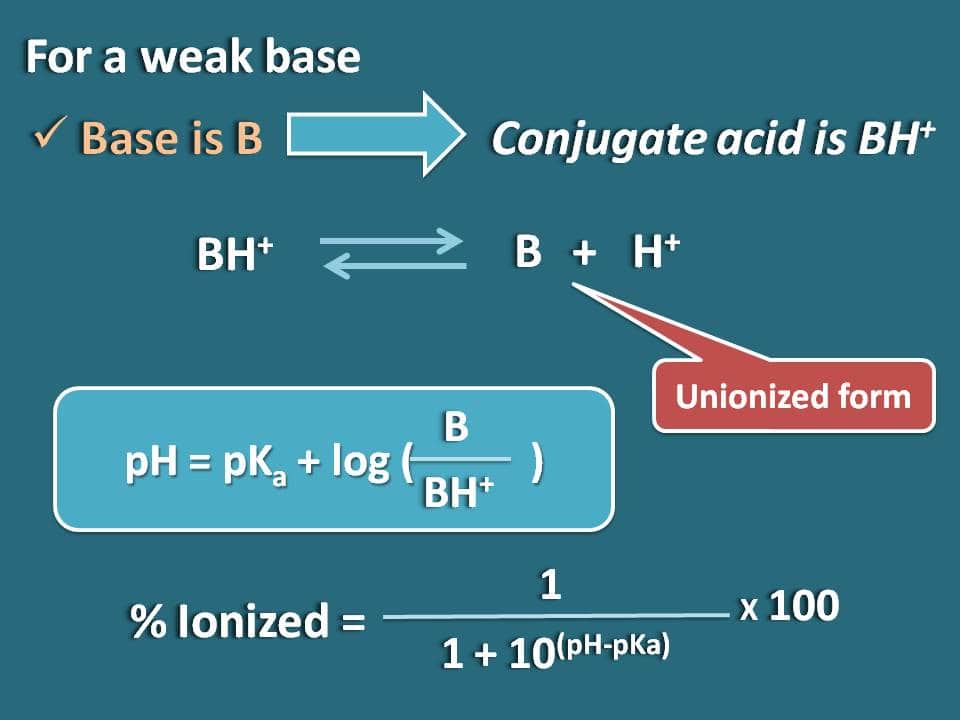

![Solved Percent ionization = [H3O+]equil/[HA}init Times 100% | Chegg.com Solved Percent ionization = [H3O+]equil/[HA}init Times 100% | Chegg.com](https://d2vlcm61l7u1fs.cloudfront.net/media%2F111%2F1110fa34-0985-4a45-9e25-717b01b554f8%2Fimage)