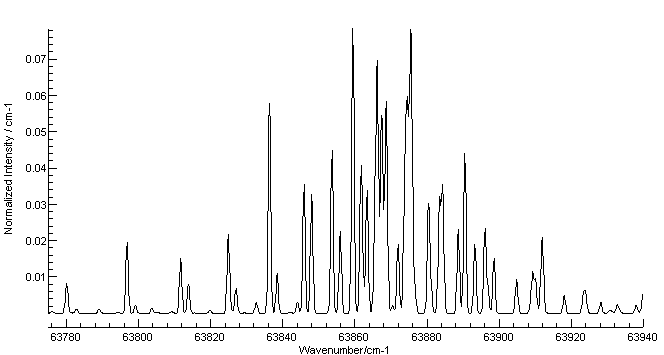
Energy spectrum of the electrons emitted in the ionization of ammonia... | Download Scientific Diagram

Study of Atmospheric Pressure Chemical Ionization Mechanism in Corona Discharge Ion Source with and without NH3 Dopant by Ion Mobility Spectrometry combined with Mass Spectrometry: A Theoretical and Experimental Study | The

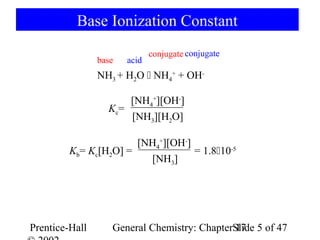
Liquid ammonia ionises to a slight extent. At `-50^(@)C`, its self ionisation constant, `K_(NH_(3))= - YouTube
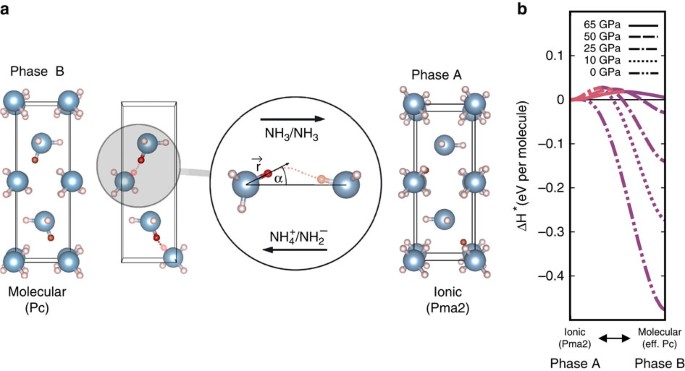
Ammonia as a case study for the spontaneous ionization of a simple hydrogen-bonded compound | Nature Communications

Quantitative Operando Detection of Electro Synthesized Ammonia Using Mass Spectrometry - Krempl - 2022 - ChemElectroChem - Wiley Online Library
![Liquid ammonia ionizes to slight extent. At - 50^∘C , its self - ionization constant, K = [NH4^+] [NH2^-] = 10^-30 M^2 . How many amide ions are present per ml of pure liquid ammonia? ( NA = 6 × 10^23 ) Liquid ammonia ionizes to slight extent. At - 50^∘C , its self - ionization constant, K = [NH4^+] [NH2^-] = 10^-30 M^2 . How many amide ions are present per ml of pure liquid ammonia? ( NA = 6 × 10^23 )](https://haygot.s3.amazonaws.com/questions/1946954_1680835_ans_ea35f931269d4fc69c74338ac2033af8.jpg)
Liquid ammonia ionizes to slight extent. At - 50^∘C , its self - ionization constant, K = [NH4^+] [NH2^-] = 10^-30 M^2 . How many amide ions are present per ml of pure liquid ammonia? ( NA = 6 × 10^23 )
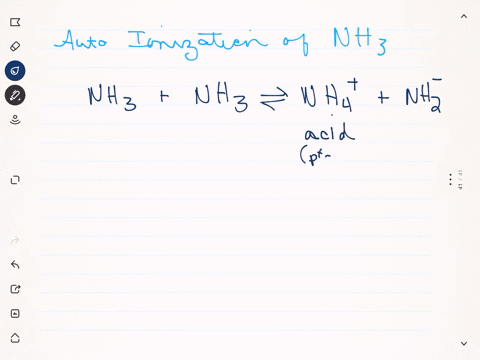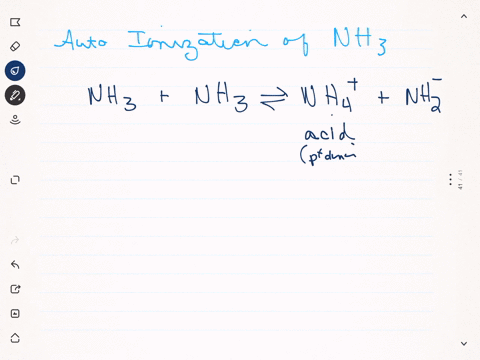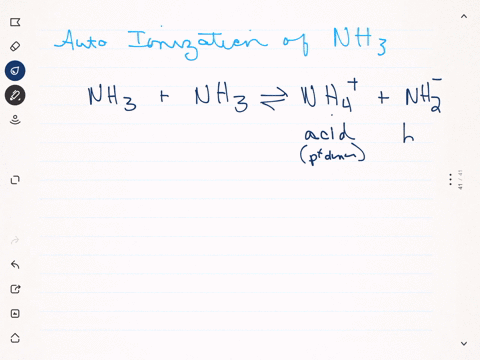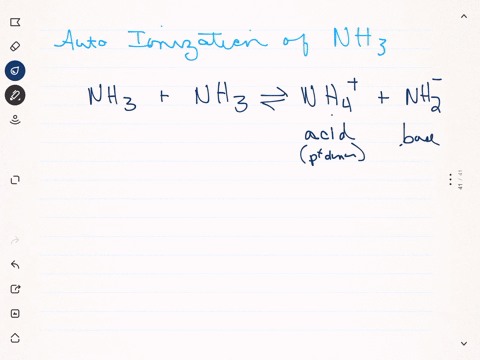
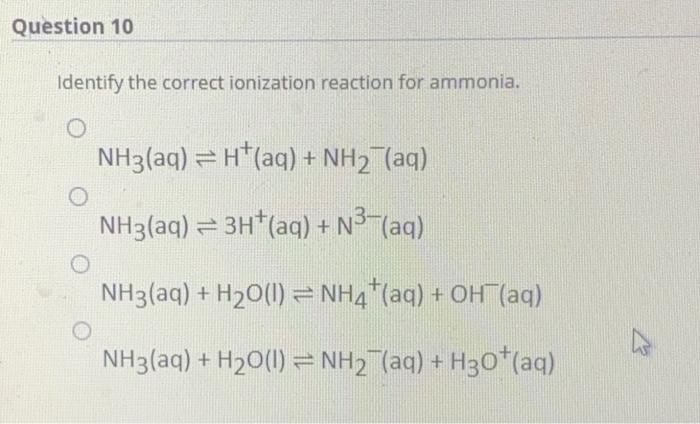
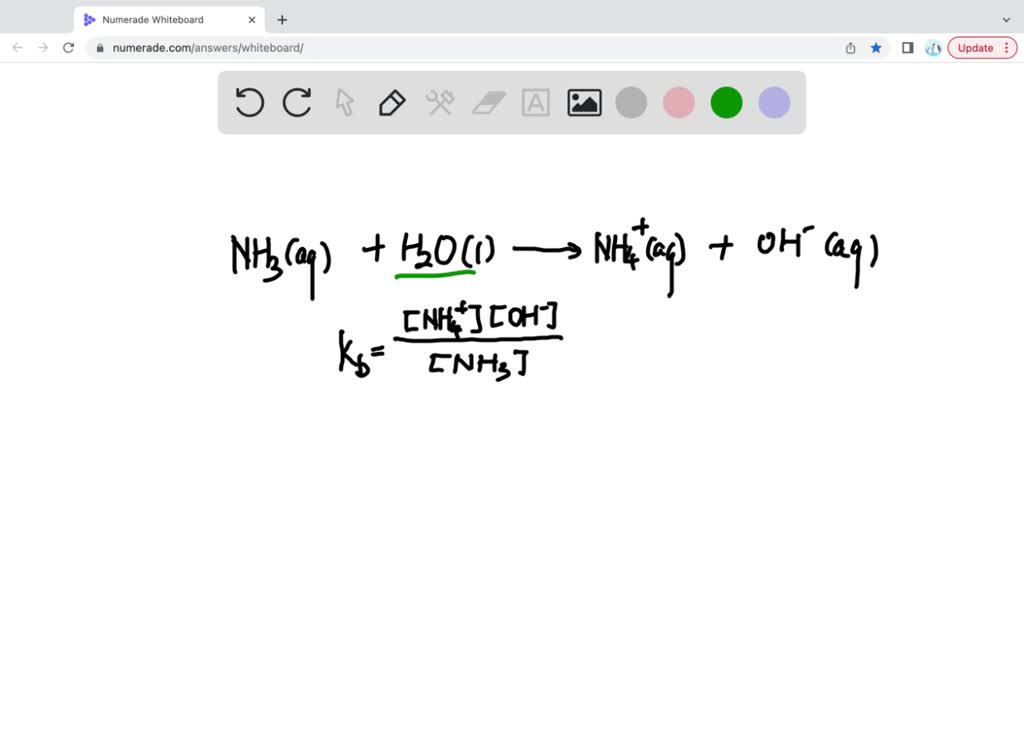
SOLVED: Write the ionization reaction for ammonia in water: Why is NH; considered "weak" base? Which is the stronger base, ammonia or hydroxide ion? Explain how you know.
![Liquid ammonia ionizes to slight extent. At - 50^∘C , its self - ionization constant, K = [NH4^+] [NH2^-] = 10^-30 M^2 . How many amide ions are present per ml of pure liquid ammonia? ( NA = 6 × 10^23 ) Liquid ammonia ionizes to slight extent. At - 50^∘C , its self - ionization constant, K = [NH4^+] [NH2^-] = 10^-30 M^2 . How many amide ions are present per ml of pure liquid ammonia? ( NA = 6 × 10^23 )](https://dwes9vv9u0550.cloudfront.net/images/11548439/2313bf3e-ac46-4626-8ef0-7c7a53c9fba2.jpg)