Octasa 400 mg Modified Release Tablets (Active substance: mesalazine) used to treat mild to moderate attacks of ulcerative colitis Stock Photo - Alamy
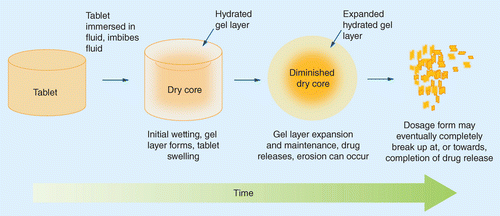
Advances in mechanistic understanding of release rate control mechanisms of extended-release hydrophilic matrix tablets | Therapeutic Delivery
![WATSONS, Trimetazidine (as Dihydrochloride) 35mg 1 Modified-Release Tablet [PRESCRIPTION REQUIRED] | Watsons Philippines WATSONS, Trimetazidine (as Dihydrochloride) 35mg 1 Modified-Release Tablet [PRESCRIPTION REQUIRED] | Watsons Philippines](https://api.watsons.com.ph/medias/prd-front-50002295.jpg?context=bWFzdGVyfGltYWdlc3wzMDcwMzF8aW1hZ2UvanBlZ3xoYjQvaGQ5Lzk0OTM2MzQ2Nzg4MTQvV1RDUEgtNTAwMDIyOTUtZnJvbnQuanBnfDdjY2RlOTllNDJjZjgwMzVkZTUxZDc3ZmY4M2E3YTA5NjhhNDc3OWY1ZjkyM2ExM2YxY2E4OWMwMzRmZmY2MTI)
WATSONS, Trimetazidine (as Dihydrochloride) 35mg 1 Modified-Release Tablet [PRESCRIPTION REQUIRED] | Watsons Philippines

Trimzed Allopathic Trimetazidine Dihydrochloride Modified Release Tablets 60mg at Rs 200/strip in New Delhi

Trimetazidine Hydrochlorides Modified Release Tablets 35mg (Taj Pharma) - Taj Generics Pharmaceuticals | Taj Pharma

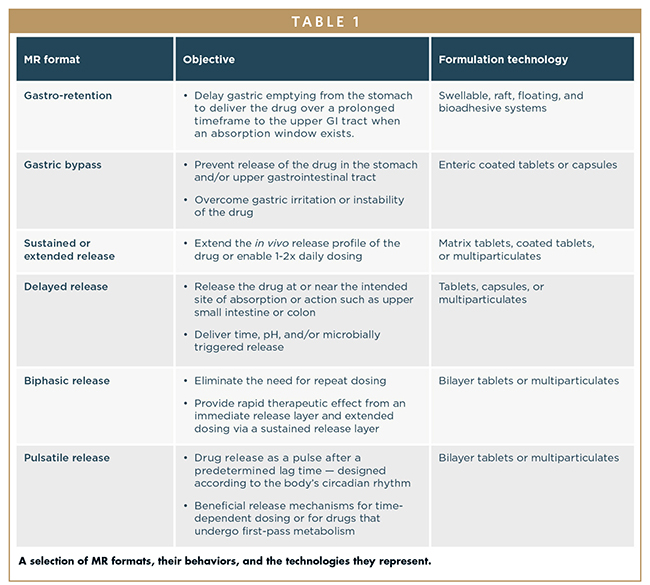
Table 4 from Oral Sustained Release Tablets: An Overview with a special emphasis on Matrix Tablet | Semantic Scholar
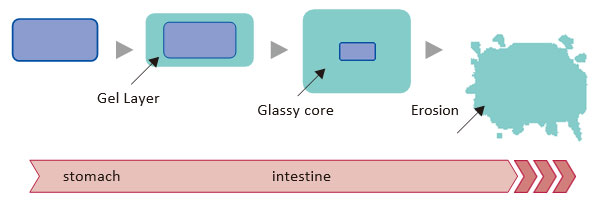
Modified and sustained release technologies: Sustained release tablets (Matrix, Gastric retentive formulation) | SPERA PHARMA, Inc.

Doxofylline Modified Release Tablets at Rs 1250/box | Pharmaceutical Tablet in Hapur | ID: 23871204291