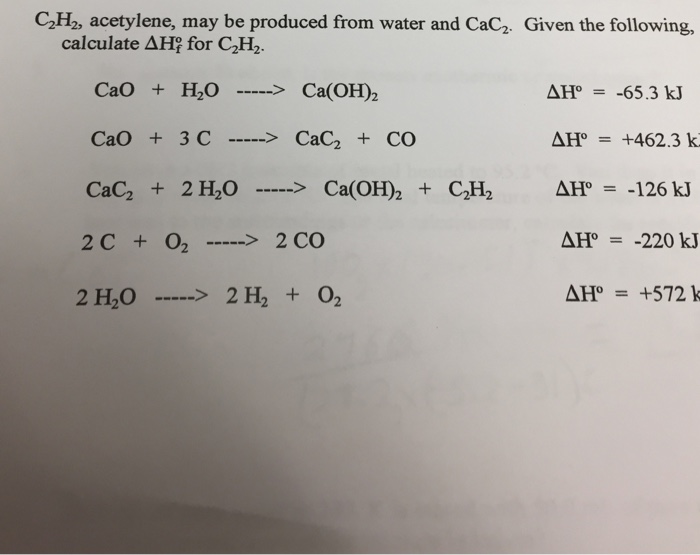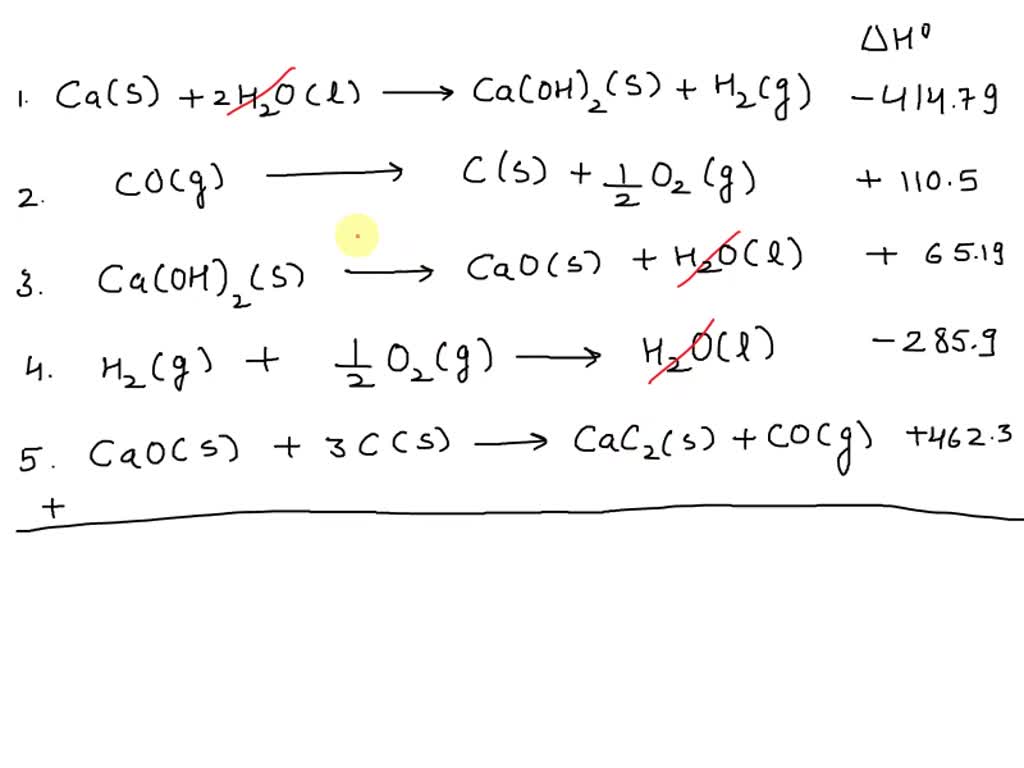
SOLVED: Calculate the standard heat of formation of calcium carbide, CaC2(s), in kJ/mol using the following thermochemical equations. Ca(s) +2H2O(l) 🡪 Ca(OH)2(s) +H2(g) ∆H° = - 414.79 kJ 2C(s) +O2(g) 🡪 2CO(g)

SOLVED:Given the thermochemical expression CaO(s)+3 C(s) ⟶CaC2( s)+CO(g) Δr H^∘ =464.8 kJ / mol (a) 2 mol O2 reacts (b) 0.115 mol N2 O3 forms (c) 4.73 g NO reacts calculate the

Refer to the following potential energy diagram and the choices above,What is the H of the reaction to form CO from C + O2

SOLVED: If 1.0X 10 kJ of energy was absorbed during the reaction, how many grams of calcium carbide (MW = 64.10 glmol) were produced given Cao + 3 € CaC2 + CO

Calcium Carbide: A Unique Reagent for Organic Synthesis and Nanotechnology - Rodygin - 2016 - Chemistry – An Asian Journal - Wiley Online Library

SOLVED: 19, Calcium oxide, CaO(s),reacts with carbon as shown: CaO(s) + 3C(s) + 462.3 k) 7 CaCz(s) + CO(g) a) Ifthe enthalpy change for a reaction is +246.7 kJ; what mass of

From the following reactions at 298 K .(A) CaC2(s) + 2H2O(l) → Ca(OH)2(s) + C2H2 (g); Δ H^∘ = - 127.9 kJ mol^-1 (B) Ca(s) + 12 O2(g) → CaO(s) ; Δ