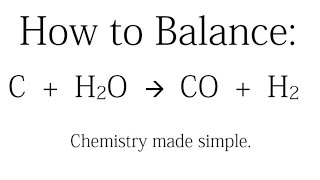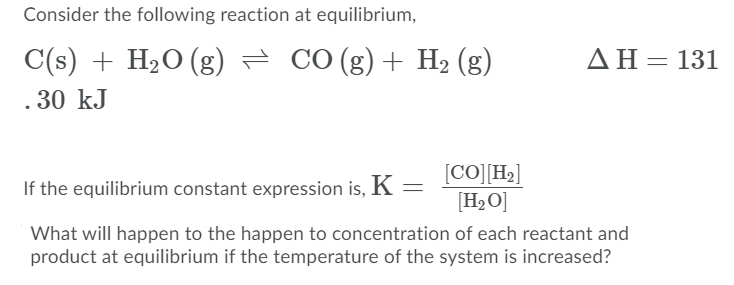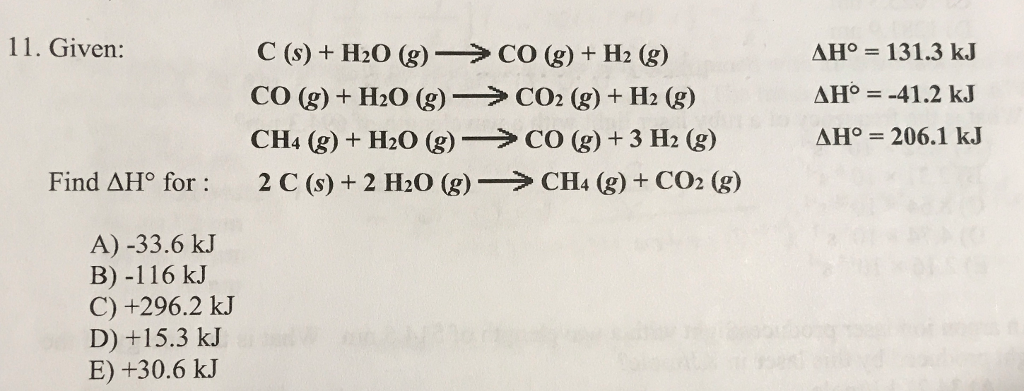C(s) +H2O(g) hArr CO(g) +H2(g) , DeltaH lt 0 the above equilibrium will proceed in forward direction when

SOLVED: How many grams of H2 would be formed if 34 grams of carbon reacted with an unlimited amount of H2O? The reaction is C + H2O → CO + H2 The
The equilibrium constant kp for the reaction H2g) + CO2,g) gives H2O(g) CO(g) is 4.0 at 1660^° C. Initially0.80 mole H2, and 0.80 mole CO2 are injected into a 5.0 liter flask.

Reaction of CO, H2O, H2 and CO2 on the clean as well as O, OH and H precovered Fe(100) and Fe(111) surfaces - Catalysis Science & Technology (RSC Publishing)

Given the reaction : C(s)+H2O(l)toCO(g)+H2(g) Calculate the volume of the gases prodcued at STP from 48.0 g of carbon.

Question Video: Identifying the Chemical Formula of the Substance Produced When Carbon Dioxide Dissolves in Water | Nagwa
Kc for CO(g) +H2O(g) ⇌ CO2(g) +H2(g) at 986°C is 0.63. A mixture of 1 mole H2O(g) - Sarthaks eConnect | Largest Online Education Community

Use the problem below to answer the question: 34 grams of carbon reacted with an unlimited amount of H20. - Brainly.com
![Calculate Kp for the reaction, C(s) + H2O(g) CO(g) + H2(g) at 990 K if the equilibrium concentration are as follows : [H2O] = 1.10 M, [CO] = [H2] = 0.2 M, Calculate Kp for the reaction, C(s) + H2O(g) CO(g) + H2(g) at 990 K if the equilibrium concentration are as follows : [H2O] = 1.10 M, [CO] = [H2] = 0.2 M,](https://dwes9vv9u0550.cloudfront.net/images/6653152/88b9752c-8929-4745-bc8f-8705cb2ad9b9.jpg)