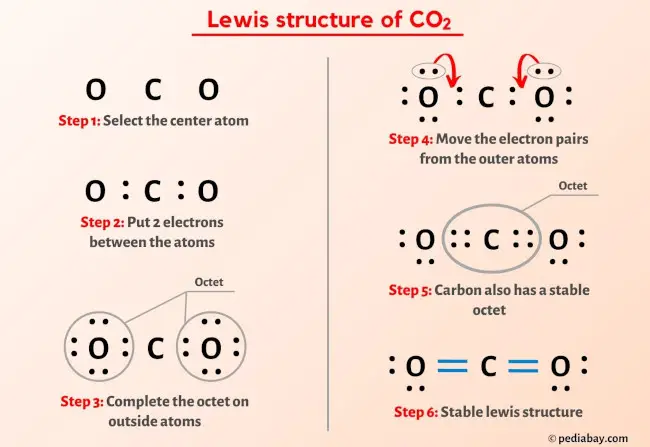
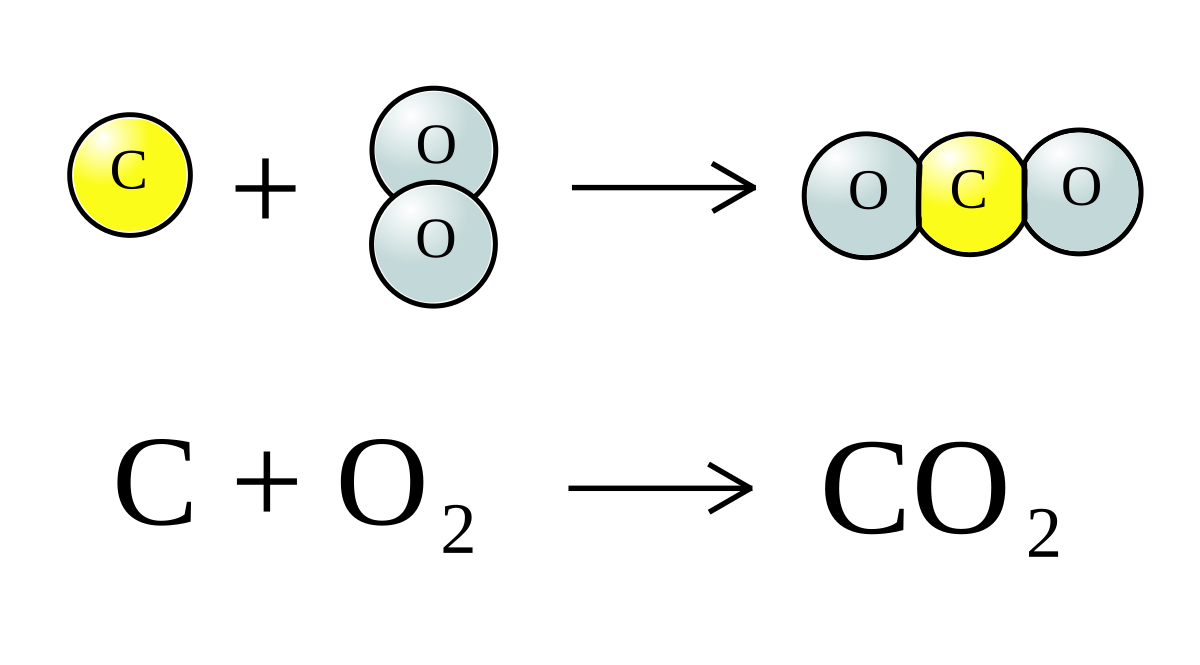
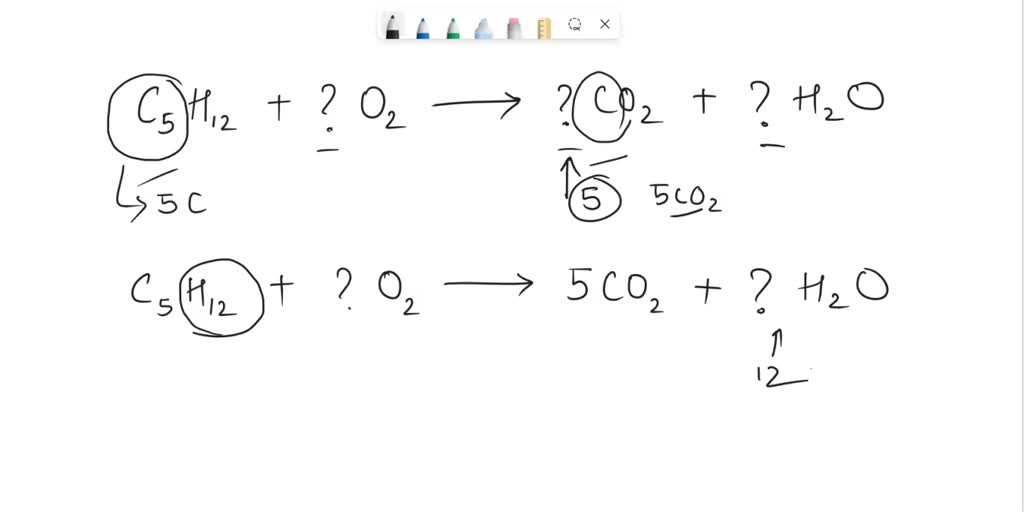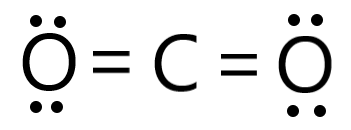22g of CO2 at 27 celsius is mixed in a closed container with 16 gram of O2 at 37 Celsius if both gases are considered as Ideal kinetic theory of gases then

Direct C–C Coupling of CO2 and the Methyl Group from CH4 Activation through Facile Insertion of CO2 into Zn–CH3 σ-Bond | Journal of the American Chemical Society
![CO2 = CO + [O]: recent advances in carbonylation of C–H bonds with CO2 - Chemical Communications (RSC Publishing) CO2 = CO + [O]: recent advances in carbonylation of C–H bonds with CO2 - Chemical Communications (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/D0CC00547A)
CO2 = CO + [O]: recent advances in carbonylation of C–H bonds with CO2 - Chemical Communications (RSC Publishing)
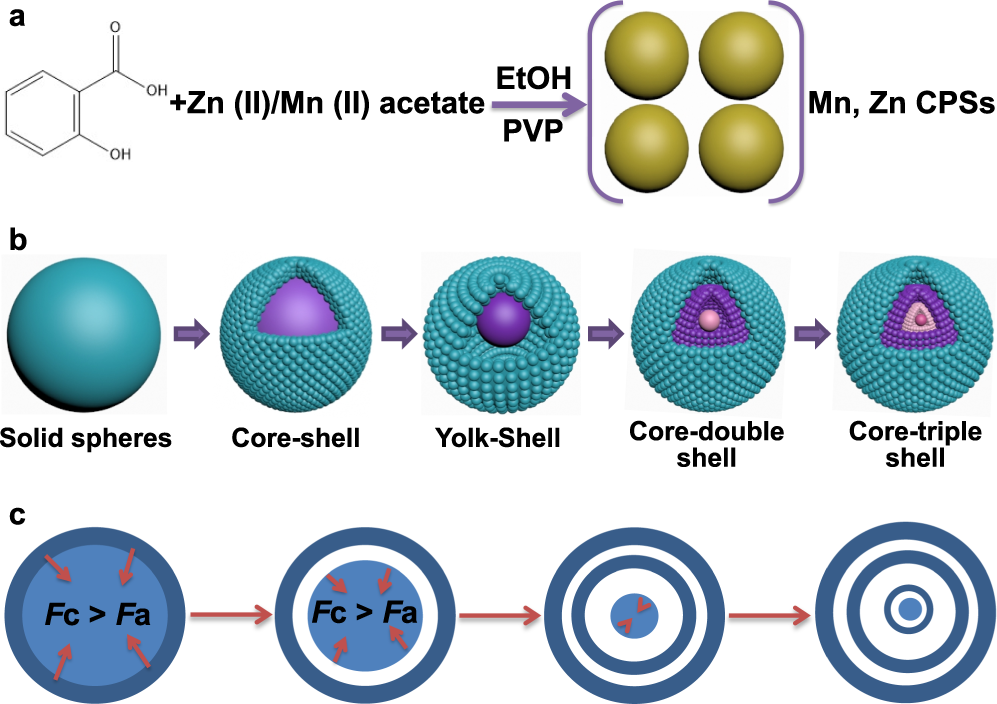
Sustained CO2-photoreduction activity and high selectivity over Mn, C-codoped ZnO core-triple shell hollow spheres | Nature Communications

For the reaction, C (s) + CO2 (g) 2CO (g) , the partial pressures of CO2 and CO are 2.0 and 4.0 atm respectively at equilibrium. The Kp for the reaction is:
Heat of reaction for, CO(g) + 1/2 O2(g)→ CO2(g)at constant V is 67.71 K cal at 17^° C. The heat of reaction at constant P at 17^° C is

Solve this: â ‹Q3 For the equilibrium C(s) + CO2(g) ⇌2CO(g) KP = 63 atm at 1000 K - Chemistry - Chemistry in Everyday Life - 11997217 | Meritnation.com

Rasprodaja Detektor Kvalitete Zraka Zidni Monitor CO2 Mjerač Temperature I Vlage U Prostoru Detektor Kvalitete Zraka CO2 Za Kućni Ured C \ Mjernih i Analitičkih instrumenata ~ Masazerzalice.com.hr

Given that C+O(2)rarrCO(2),DeltaH^(@)=-xKJ and 2CO+O(2)rarr2CO(2),DeltaH^(@)=-yKJ The enthalpy of formation of carbon monoxide will be

Atomically Dispersed Indium‐Copper Dual‐Metal Active Sites Promoting C−C Coupling for CO2 Photoreduction to Ethanol - Shi - 2022 - Angewandte Chemie International Edition - Wiley Online Library